Parachute Experiment: "Save The Egg"How big should a parachute be to safely land a raw egg from the second floor?
|
Puff MobileWhat is the best way of making a puff mobile from straw, lifesavers candy, paper clips, a meter of tape, and one sheet of paper? Make sure to take friction, air resistance, and aerodynamics into consideration!
|
MomentsNo, not that kind of "moment" that blossoms into love! We're talking about torque or turning force. Think about seesaws (or teeter totters) and how to balance them. Remember, moment = F(d) where "F" is force and "d" is distance from pivot.
EXTRACTING DNA FROM FRUITWhat can be more amazing than being able to extract the very thing that dictates who we are? Yes, DNA...the blueprint of every living thing. For this lab, we are extracting DNA from a Kiwi fruit. Of course we can also extract it from human cheek cells -- but, it is just so much fun mashing the fruit than scrubbing the inside of your mouth!
|
Balloon ExperimentJust how strong is air pressure? Well, let's see! Do you think 60 blown up balloons can support the weight of a lab table, an entire Y9 class, and the teacher?
|
EXTRACTING DNA FROM FRUIT by Kin Yuen 9C
Extracting DNA lab Report
Teacher: Mrs Nazareno
Group Members: Regina and Kin Yuen (Me!)
Date of experiment: 30-3-2011 Wednesday Day 1
Introduction
What makes you you? What makes Derek so arrogant(Oops!)? The answer is small. Yes, literally, the DNA that contains all the code of the human body is so small that need to use a microscope to see it. Think of your body as the hardware (your screen, mouse etc.) and your DNA as the software (Windows, Mac etc.). DNA stands for DeoxyRiboNucleic Acid, contains what determine who you are (Hair color , eye color etc.). In this experiment we are going to extract DNA from fruit to take a look at this acid that determines the life of trillion of beings around the globe (Not only humans have DNA – Animals and Plants too!)
What is DNA made up from?
Adenine (C5H5N5)
Cytosine (C4H5N3O)
Guanine (C5H5N5O)
Thymine (C5H6N2O2)
Learning Goal
To observe first hand that DNA is in the food we eat
To learn the simple method of DNA extraction
To explain the reason behind each step
Use of the microscope
Learning Outcomes
Able to successfully extract DNA from fruit
Able to see the extracted DNA using a microscope
Materials (Part 1)
Kiwi fruit
Ziplock bag
250ml concial flask
Strainer
Funnel
2 teaspoons of detergent
Test tube rack
Boiling tube
10ml cold ethanol
Measuring cylinder
Spatula
Boiling tube rack
Procedure (Part 1)
1. Put the fruit in the ziplock bag. Remove as much air as you can before sealing the bag.
2. Mash up the fruit with your hands and fingers for 2 minutes. Be careful not to break your bag.
3. Add 2 teaspoons of detergent to the bad and zip it closed. Mash again for 1 minute.
4. Carefully pour the mush into the strainer. Let the liquid drip through into the conical flask.
5. After most of the liquid has dripped into the beaker, carefully remove the strainer with the mush and throw it in the trash. Transfer the contents of the conical flask into the boiling tube.
6. Tilt the boiling tube slightly and gently pour in the 10ml of ethanol, letting it slowly pour down the side of the tube. DO NOT MIX!
7. Observe and wait a while. You will see the DNA start to collect as a goopy glob, and you can take it out it using a spatula.
Materials (Part 2)
Microscope
Slide
Slide cover
Methylene Blue
DNA extracted from Part 1
Spatula
Procedure (Part 2)
1. Put away all materials, leaving only the boiling tube rack, boiling tube, and spatula.
2. Get a microscope, slide, slide cover, and methylene blue stain.
3. Using the spatula, scoop up some DNA and and put it on the slide.
4. Add a drop of blue stain, and put the slide cover on.
5. Use a paper towel to suck off any excess stain.
6. Examine the DNA using a microscope.
7. Call Mrs Naz once you have a good view of the DNA.
8. Take a picture which will be included in your lab report (Which is the document you are reading now).
Results
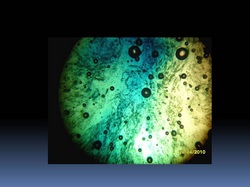
A strip of DNA – Mission success! (look at photo on top)
[note: this is not the actual photo from Kin Yuen's group, but I couldn't download the original photo from his report...anyway, DNA is DNA...]
Frequently Asked Questions
Q: What is DNA?
A: As mentioned in the introduction, DNA stands for Deoxyribonucleic Acid, which carries genetic information in all living things that is needed for the function or development of that being. You may compare it to a blueprint or recipe, which are similar in function to the DNA. DNA is inherited from your parents (Half in sperm cells from dad, half in egg cells from mum), so that's why we share traits with our parents (Eye color, hair color etc.).
Q: Why is it important to mash the fruit very well?
A: We need to smash well to get the DNA out from the nucleus and into the liquid that will drip into the conical flask.
Q: How does adding the detergent help release the DNA?
A: There is a enzyme in the detergent which is used to clean stuff, but when you add it to the juice-mixture it breaks down the cell wall of the kiwi fruit cells, which helps to release the DNA from our examination.
Q: Why do we add cold ethanol?
A: Firstly, you must add cold ethanol because there is no hot ethanol. Ethanol evaporates very easily so must be kept cold to prevent it from fleeing. The ethanol suspends the DNA onto the surface for easy collection.
Q: Why do you have to add stain to the DNA slide?
A: The DNA is hard to see as it is a transparent-translucent blob. Adding the stain will add color to it for better viewing.
Teacher: Mrs Nazareno
Group Members: Regina and Kin Yuen (Me!)
Date of experiment: 30-3-2011 Wednesday Day 1
Introduction
What makes you you? What makes Derek so arrogant(Oops!)? The answer is small. Yes, literally, the DNA that contains all the code of the human body is so small that need to use a microscope to see it. Think of your body as the hardware (your screen, mouse etc.) and your DNA as the software (Windows, Mac etc.). DNA stands for DeoxyRiboNucleic Acid, contains what determine who you are (Hair color , eye color etc.). In this experiment we are going to extract DNA from fruit to take a look at this acid that determines the life of trillion of beings around the globe (Not only humans have DNA – Animals and Plants too!)
What is DNA made up from?
Adenine (C5H5N5)
Cytosine (C4H5N3O)
Guanine (C5H5N5O)
Thymine (C5H6N2O2)
Learning Goal
To observe first hand that DNA is in the food we eat
To learn the simple method of DNA extraction
To explain the reason behind each step
Use of the microscope
Learning Outcomes
Able to successfully extract DNA from fruit
Able to see the extracted DNA using a microscope
Materials (Part 1)
Kiwi fruit
Ziplock bag
250ml concial flask
Strainer
Funnel
2 teaspoons of detergent
Test tube rack
Boiling tube
10ml cold ethanol
Measuring cylinder
Spatula
Boiling tube rack
Procedure (Part 1)
1. Put the fruit in the ziplock bag. Remove as much air as you can before sealing the bag.
2. Mash up the fruit with your hands and fingers for 2 minutes. Be careful not to break your bag.
3. Add 2 teaspoons of detergent to the bad and zip it closed. Mash again for 1 minute.
4. Carefully pour the mush into the strainer. Let the liquid drip through into the conical flask.
5. After most of the liquid has dripped into the beaker, carefully remove the strainer with the mush and throw it in the trash. Transfer the contents of the conical flask into the boiling tube.
6. Tilt the boiling tube slightly and gently pour in the 10ml of ethanol, letting it slowly pour down the side of the tube. DO NOT MIX!
7. Observe and wait a while. You will see the DNA start to collect as a goopy glob, and you can take it out it using a spatula.
Materials (Part 2)
Microscope
Slide
Slide cover
Methylene Blue
DNA extracted from Part 1
Spatula
Procedure (Part 2)
1. Put away all materials, leaving only the boiling tube rack, boiling tube, and spatula.
2. Get a microscope, slide, slide cover, and methylene blue stain.
3. Using the spatula, scoop up some DNA and and put it on the slide.
4. Add a drop of blue stain, and put the slide cover on.
5. Use a paper towel to suck off any excess stain.
6. Examine the DNA using a microscope.
7. Call Mrs Naz once you have a good view of the DNA.
8. Take a picture which will be included in your lab report (Which is the document you are reading now).
Results
A strip of DNA – Mission success! (look at photo on top)
[note: this is not the actual photo from Kin Yuen's group, but I couldn't download the original photo from his report...anyway, DNA is DNA...]
Frequently Asked Questions
Q: What is DNA?
A: As mentioned in the introduction, DNA stands for Deoxyribonucleic Acid, which carries genetic information in all living things that is needed for the function or development of that being. You may compare it to a blueprint or recipe, which are similar in function to the DNA. DNA is inherited from your parents (Half in sperm cells from dad, half in egg cells from mum), so that's why we share traits with our parents (Eye color, hair color etc.).
Q: Why is it important to mash the fruit very well?
A: We need to smash well to get the DNA out from the nucleus and into the liquid that will drip into the conical flask.
Q: How does adding the detergent help release the DNA?
A: There is a enzyme in the detergent which is used to clean stuff, but when you add it to the juice-mixture it breaks down the cell wall of the kiwi fruit cells, which helps to release the DNA from our examination.
Q: Why do we add cold ethanol?
A: Firstly, you must add cold ethanol because there is no hot ethanol. Ethanol evaporates very easily so must be kept cold to prevent it from fleeing. The ethanol suspends the DNA onto the surface for easy collection.
Q: Why do you have to add stain to the DNA slide?
A: The DNA is hard to see as it is a transparent-translucent blob. Adding the stain will add color to it for better viewing.
A LITTLE STUDENT WIT!
Who says lab reports can't be fun? Here is a little taste of student wit from Kin Yuen of 9C as he answers questions about rusting.
Conclusion, Discussion, Explanation......
I know you have a lot of questions, I know. Let me do it one by one.
Q: Why do we need to seal it with parafilm? from Sara Film
A: We need to seal with parafilm, Sara Film, is because we need to make a closed system. What is a closed system? A closed system means a system that sealed so that nothing can get in or escape during the reaction. It is important so that nothing can get in and affect our results.
Q: Why would the nail rust in water anyway? from Dis Liek Wusting
A: I know you dislike rusting, Dis Liek Wusting, but it is a natural phenomenon. The nail rusts in test tube 1,2,3 is because of water, as you said. But why? Actually iron is a quite reactive metal, and it reacts with oxygen most of the time. When it reacts with oxygen, a brown substance which is called iron oxide is made, what is commonly called rust. So all that rust you see is iron oxide. Remember, water contains oxygen (H2O) so it can react with the iron nail!
Q: Why the nail in don't test tube 4 rust? from Kelshium Clorime
A: Test tube 4 doesn't rust it contains calcium chloride, Kelshium Clorime. The calcium chloride is a drying agent and it absorbs all the water vapor in the air, leaving no water to react with the nail, thus the nail doesn't rust.
Conclusion, Discussion, Explanation......
I know you have a lot of questions, I know. Let me do it one by one.
Q: Why do we need to seal it with parafilm? from Sara Film
A: We need to seal with parafilm, Sara Film, is because we need to make a closed system. What is a closed system? A closed system means a system that sealed so that nothing can get in or escape during the reaction. It is important so that nothing can get in and affect our results.
Q: Why would the nail rust in water anyway? from Dis Liek Wusting
A: I know you dislike rusting, Dis Liek Wusting, but it is a natural phenomenon. The nail rusts in test tube 1,2,3 is because of water, as you said. But why? Actually iron is a quite reactive metal, and it reacts with oxygen most of the time. When it reacts with oxygen, a brown substance which is called iron oxide is made, what is commonly called rust. So all that rust you see is iron oxide. Remember, water contains oxygen (H2O) so it can react with the iron nail!
Q: Why the nail in don't test tube 4 rust? from Kelshium Clorime
A: Test tube 4 doesn't rust it contains calcium chloride, Kelshium Clorime. The calcium chloride is a drying agent and it absorbs all the water vapor in the air, leaving no water to react with the nail, thus the nail doesn't rust.
